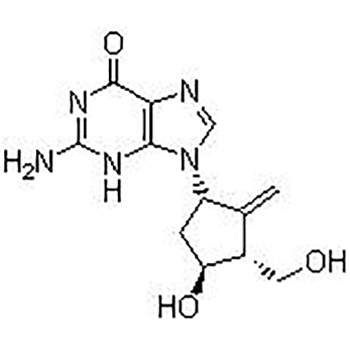
High reputation Sulfamonomethoxine 1220-83-3 - Rivaroxaban(S)-Rivaroxaban;BAY 59-7939;RivaroxabanIsomer;Rivaroxaban; – JIN DUN
High reputation Sulfamonomethoxine 1220-83-3 - Rivaroxaban(S)-Rivaroxaban;BAY 59-7939;RivaroxabanIsomer;Rivaroxaban; – JIN DUN
High reputation Sulfamonomethoxine 1220-83-3 - Rivaroxaban(S)-Rivaroxaban;BAY 59-7939;RivaroxabanIsomer;Rivaroxaban; – JIN DUN Detail:
Used in adult patients with elective hip or knee replacement surgery to prevent venous thrombosis (VTE).
It is used to prevent or reduce the formation of thrombosis, deep vein thrombosis (DVT) and pulmonary embolism (PE) after hip or knee replacement surgery.
The recommended dose is oral rivaroxaban 10mg, once a day. Rivaroxaban 10mg can be taken with food or alone. Rivaroxaban 15mg or 20mg tablets should be taken with food.
standard: usp40
Assay: 99-102%
Exterior: White powder
Package : 25kg/drum
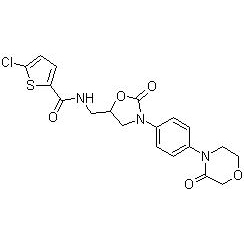
Take 4-(4-aminophenyl) morpholin-3-one (SM-1), (S)-N-glycidyl phthalimide (SM-2), 5-chlorothiophene-2- Formyl chloride (SM-3) is the starting material. After condensation, cyclization, aminolysis and acylation, the crude rivaroxaban is obtained, and the finished rivaroxaban is refined.
Preparation: This product is a red film-coated tablet.
Accessories:
Tablet core: microcrystalline cellulose, croscarmellose sodium, lactose monohydrate, hypromellose, sodium lauryl sulfate, magnesium stearate
Coating: polyethylene glycol, hypromellose, titanium dioxide, iron oxide red
Rivaroxaban is a highly selective, oral drug that directly inhibits factor Xa. Through highly selective and competitive inhibition of free and bound factor Xa and prothrombin activity, prolong the activated partial thromboplastin time plate (PT) and prothrombin time (aPTT) in a dose-dependent manner, which can interrupt the coagulation waterfall The endogenous and exogenous pathways inhibit the production of thrombin and thrombosis. Rivaroxaban does not inhibit thrombin (activating factor Ⅱ), nor has it proven to have an effect on platelets
The essential difference between rivaroxaban and fondaparinux/heparin is that it does not require the participation of antithrombin III and can directly antagonize free and bound factor Xa; while heparin requires antithrombin III to function, and It has no effect on factor Xa in the prothrombin complex.
Product detail pictures:
Related Product Guide:
Bear "Customer initially, High-quality first" in mind, we work closely with our prospects and supply them with efficient and specialist companies for High reputation Sulfamonomethoxine 1220-83-3 - Rivaroxaban(S)-Rivaroxaban;BAY 59-7939;RivaroxabanIsomer;Rivaroxaban; – JIN DUN , The product will supply to all over the world, such as: Jersey, Lebanon, Luxembourg, With the advanced workshop, professional design team and strict quality control system, based on mid- to high-end marked as our marketing positioning, our products are fast selling onto European and American markets with our own brands such as below Deniya, Qingsiya and Yisilanya.
Speaking of this cooperation with the Chinese manufacturer, I just want to say"well dodne", we are very satisfied.


![Competitive Price for Crystal Glue Mold Set - 2-butyl-5-nitro-3-benzofuranyl)[4-[3-(dibutylaMino)propoxy]phenyl] – JIN DUN](https://cdn.globalso.com/jindunchem-med/922e79ba.jpg)
![One of Hottest for Salicylic Acid - Casp ungin Acetate;Caspofungin acetate;Cancidas;Caspofungin acetate [USAN:BAN:JAN]; – JIN DUN](https://cdn.globalso.com/jindunchem-med/fbe17385.jpg)